
We will achieve the SDGs through “cell manufacturing.

Understanding the origins of human tissue and building technologies to nurture it.
Regenerative medicine consists of a series of processes: collection of human cells (especially patient’s cells), in vitro cell amplification culture, tissue culture, and transplantation to the diseased area. We are researching methods for designing human stem cell cultures.

- What are iPS cells?
-
When a specific group of factors is introduced into cells in the body, the cells become rejuvenated and diverse. Pluripotent stem cells artificially created in this way are called iPS cells.
- What is regenerative medicine?
-
A medical treatment that aims to restore lost human body functions by regenerating organs and tissues that are defective, dysfunctional, or inadequate using stem cells.
- What does regenerative medicine expect from iPS cells?
-
iPS cells are expected to be used in regenerative medicine because of their pluripotency and ability to proliferate and differentiate into various organs and cells.
 Prof.KINO-OKA
Prof.KINO-OKAWe are learning about the development of human stem cells with pluripotent to induce differentiation and developing a system to evaluate cell maturity to analyze cell shape and color.


Automation of the tissue culture production process
In recent years, in the field of transplantation therapy, cells have been cultured in vitro and then differentiated into tissues and organs, which are eventually transplanted and reconstructed in the patient.
- What is in vitro ?
-
A test to detect drug reactions by artificially creating an environment similar to that in the body using human or animal tissues in test tubes or incubators.



We are aiming to develop a “fully automated culture reactor” that can realize three-dimensional reconstruction of keratinocytes into skin tissue, a method of culture and skin production.
Research on the production process of human cultured tissue for clinical application
〇 Research on the production process of cultured cells and tissues for transplantation
〇 Product process of raw materials and transplanted cells and tissues with patient’s own cells.
〇 Development of unique features such as heterogeneity of raw materials and variation in production scale
〇 Establishment of methods for process control focusing on culture and quality control of cell and tissue products





The development of analytical techniques for quality control will enable the realization of “in silico” production that can predict patient outcomes.
- What is in silico ?
-
In silico refers to experiments using computers, which can predict and analyze the results of actual experiments and is expected to contribute to the validation of general scientific hypotheses and to events that are difficult to measure with current technology.


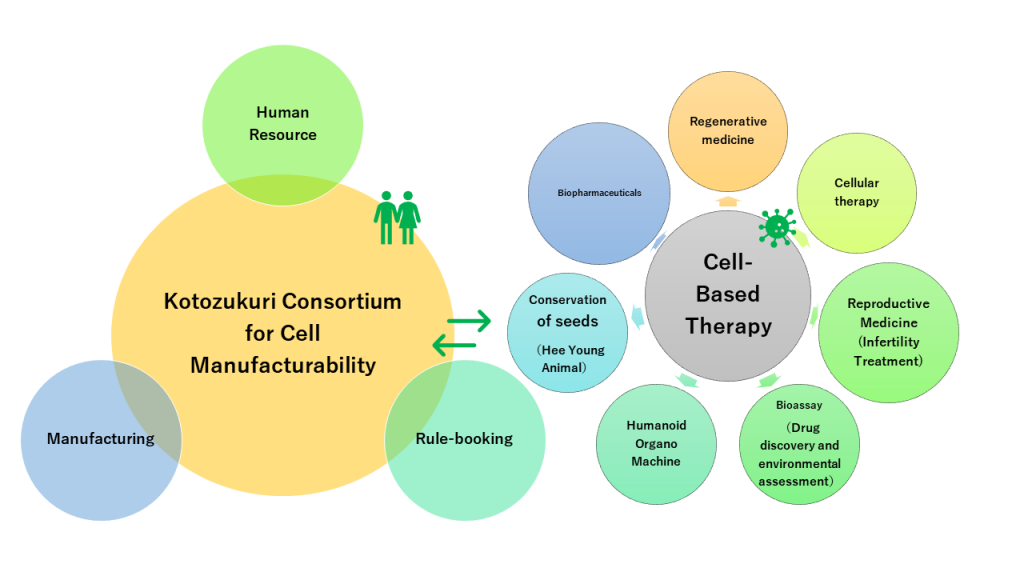
Creating human resources with good sense at the “Kotozukuri Consortium for Cell Manufacturability”
With the aim of revitalizing industry through the trinity of industry, government, and academia, we are working to create a new core by bringing together experts from different fields to collaborate and play a key role in achieving our goals.
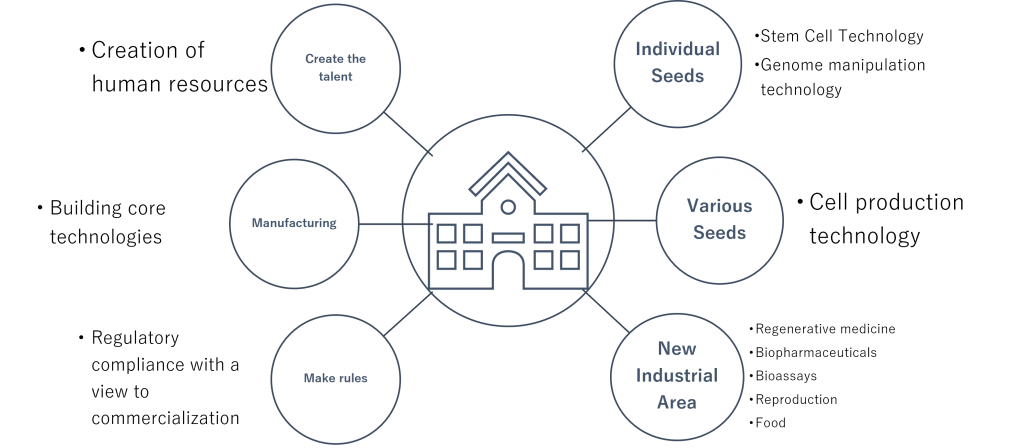




The formation of a cross-company center will lead to the creation of an ecosystem that can serve as a place for professional development, technology development, and social implementation, as well as for education and research activities.
Cultured meat reduces methane and livestock slaughter
Meat farming does not produce as much methane and can significantly reduce carbon emissions. In addition, the cost of producing cultured meat using iPS cells is relatively low and is expected to be implemented in society.


- What is cultured meat?
-
A meat substitute that artificially produces edible meat by cultivating and increasing small amounts of cells taken from domestic animals such as cattle.



The day is approaching when cultured meat will be the solution to the food problems associated with population growth.
